Soil chemistry is crucial for understanding and managing various soil processes that impact plant growth, ecosystem health, and environmental quality. It helps in optimizing nutrient availability, controlling pollutants, and understanding how soil properties influence plant growth and microbial activity. It is affected by mineral composition, organic matter and environmental factors.
One of the major factor of soil chemistry is redox, apart from pH etc.
Redox ( that is, reduction-oxidation), is the measurement of electrons and is a vital but often overlooked factor in soil chemistry. Redox (Eh) refers to the chemical processes that involve the gain or loss of oxygen and electrons.
To simplify the above definition, reduction reactions occur when an element gains electrons from another element, reducing their charge (making the charge more negative). While oxidation reactions occur when an element loses electrons.
Also, for quick remembrance of the two redox terminologies, they can be called LEO and GER.
LEO: Loss of Electrons is Oxidation
GER: Gain of Electrons is Reduction
Redox relate to the availability of nutrients such as nitrogen, phosphorus, iron, and manganese in the soil.
Most often, soil scientists focus more on soil pH rather than redox balance. But redox potential plays a critical role in soil health and function. Therefore, it can be stated that redox governs nutrient availability, microbial processes, and overall soil health. These processes are key to understanding soil health, nutrient cycling and waterlogging.
A healthy soil is being said to be a soil that maintains redox balance through water resilience, organic matter, and having living roots in the system.
THE MAJOR DRIVE OF REDOX
Redox is driven by electron transfer:
a. Reduction (gain of electrons) stores energy
b. Oxidation (loss of electrons) losses energy
When a soil becomes oxidised, nutrients becomes less soluble. While a waterlogged soils becomes overly reduced and can make nutrients toxic. Such soils depleted in oxygen, makes soil microbes to utilize other elements such as iron, manganese, nitrate, sulfate as electron acceptors.
IMPORTANCE OF SOIL CHEMISTRY
1. NUTRIENT AVAILABILITY:
Soil chemistry governs the availability of essential nutrients for plant growth. Understanding how these nutrients are stored, released, and interact with other soil components is vital for efficient fertilization and maximizing crop yields.
2. PLANT GROWTH AND REPRODUCTION:
Soil pH, a key aspect of soil chemistry, directly influences nutrient availability and plant growth. Optimizing soil pH ensures that plants can access the nutrients they need for healthy development.
3. SOIL HEALTH AND FERTILITY:
Soil chemistry plays a significant role in determining overall soil health and fertility. By understanding the chemical composition of the soil, the soil’s ability can be assessed to support plant life, retain water, and resist degradation.
4. ENVIRONMENTAL PROTECTION:
Soil chemistry is essential for monitoring and controlling pollutants in the environment. It helps in understanding how contaminants move through the soil, how they interact with other chemicals, and how to remediate contaminated sites.
5. ECOSYSTEM FUNCTIONING:
Soil chemistry is a critical indicator of wetland ecological condition and plays a role in nutrient cycling, water quality, and the overall health of various ecosystems.
6. IMPACTATION OF MODERN AGRICULTURE:
Soil chemistry helps in understanding the effects of intensive agricultural practices on soil properties and in developing strategies for sustainable land management.
7. GLOBAL WARMING EFFECTS:
Soil chemistry research is crucial for understanding the impact of climate change on soil properties and for developing strategies to mitigate the effects of global warming on soil health and food production.
8. Soil chemical properties influences countless number of soil processes. The chemicals in soil, from solid minerals to nutrients and other salts in solution, affect not only plant growth and microbial diversity but also soil greenhouse gas emissions and the transport and breakdown rate of nearly any chemical from nutrients to hazardous substances. This gives reasons why there is the need to understand soil chemistry as an important tool in the management of soils.
IMPORTANCE OF REDOX
1. Redox causes soils at depth to become grey and red in colour. For example, pit soils at a depth of about 75 to 132 cm , do show different horizontal layers of different red and grey shades running across the pit.
2. Reduction and oxidation (redox) reactions affect numerous soil processes, ultimately influencing nutrient availability and mobility, microbial activity, soil pH, and even soil colour.
3. Redox processes alter soil appearances in ways that help scientist interpret soil formation conditions.
4. Reduction and oxidation features (redox features, or redoximorphic features) are indicators of important soil conditions, particularly oxygen status.
5. In drought conditions, soil redox features can be used to avoid potentially costly issues, like basement or septic system placement in an area with regularly saturated conditions.
EXAMPLES OF REDOX REACTION
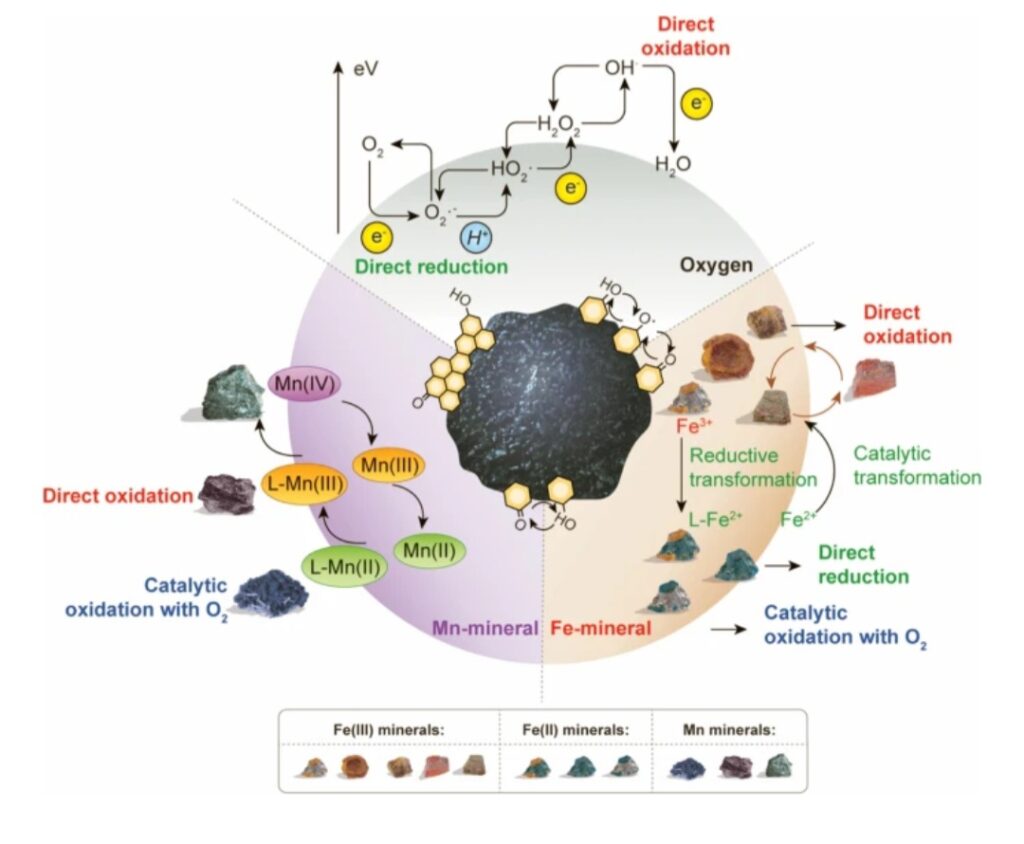
The most common elements that usually undergo redox reactions in soils are iron (Fe) and manganese (Mn). They tend to lose their electrons to elements like O in oxygen gas (O2) and N in nitrate (NO3–).
These two elements O and N are nonmetal elements that frequently gain electrons, and become “reduced”. They are called oxidizing agents, because they help oxidize other elements. Of these, oxygen is the best oxidizing agent found in nature.
On the other hand, metals like Fe and Mn, however, tend to act as reducing agents, because they help reduce the O and N.
“Rusting” phenomena is a type of redox reactions. For example, the iron found in iron ore and cast iron pots is Fe0. Fe0 readily donates electrons, as shown in this reaction:
Fe0 –> Fe2+ + 2e–
The Fe0 gave up 2 electrons, and become oxidized to Fe2+ at the products side. When FeO gave up electrons, the Fe was oxidized, and developed a 2+ charge. Fe2+ can precipitate with OH- and form green rusts. Or, it can react again as shown below:
Fe2+ –> Fe3+ + e-
From the above reactions, electrons are released. It is important to note that both oxidation reactions and reduction reactions go hand in hand. This gives the reason why some reactions are stated half reactions and the reactions will show where the electrons released will go.
In well-aerated soils, the most common oxidizing agent is O2 gas. This O2 is reduced through half reaction:
O2 + 4e- –>2O2-
or O + O + 4e- –>2O2-
From the above reaction, the two oxygens in oxygen gas have accepted 2 electrons each, for a total of 4 electrons.
Surpposing the first half reaction is doubled with iron, and then added to the one with oxygen, then a balanced reaction can be shown below:
2Fe0 + O2 –> Fe2+ + O2-
The iron has donated 2 electrons per atom, or 4 electrons to the oxygen gas. The oxygen gas develops a -2 charge, and the iron develops a +2 charge. Then, in the presence of water, Fe reacts with water to form green rust:
Fe2+ + 2H2O –> Fe(OH)2
Any Fe2+ in solution can alternatively react again with oxygen, to further oxidize the iron:
2Fe2+ + O2 + –>Fe3+ + 2O2-

REDUCTION FEATURES
Under saturated soil conditions;
i. Soil ped do show grey with tinted rusty red colour.
ii. Few plant roots may stick out of the soil
iii. Sandy soils with gleyed conditions may change colour to light blue when pulled out of the soil.
This grey colour found in the soil indicate a lack of oxygen at that soil area. It should be noted that additional factors can contribute to this grey colour apart from lack of oxygen. For example, during drought, redox colour can be developed due to regular saturation. This makes soil colour an important indicator of expected wetness conditions.
Some of the different compounds in soil that can be reduced include: nitrate, iron, manganese and sulfur etc.
OXIDATION FEATURES
Under unsaturated condition, oxidation takes place in the soil due to aboundance of oxygen in the soil. The following features can be noticed in such soils;
i. Dull gray soil dominates most of the soil area.
ii. The dull gray soil will show high contrasting red iron concentrations with few white calcium carbonate spots.
In summary, Soil showing redox concentrations will be red in colour, depletions (grey), and carbonate accumulations (white)
RELATIONSHIP BETWEEN SOIL PHASES AND SOIL CHEMICALS
The soil is made of three phases, solid, liquid and gaseous phase. These three soil phases are made up of chemicals. Therefore, it can be said that the soil is made of chemicals.
Soil contains countless numbers of chemicals which react with each other on a regular basis. These chemicals are important when discussing redox and are the main bases of the three phases of the soil.
SOIL SOLIDS
An average soil is about 50% solid particles. These solid particles are divided into mineral and organic parts. The minerals are inorganic, meaning they do not contain carbon (with the major exception of carbonates). On the other hand, the organic particles contain carbon (excluding carbonates ).
The minerals are formed from the weathering of rocks. They supply nutrients to the soil,
give the soil its structure and provide adhesive or adsorbtive surfaces for other chemicals to be held in the soil and supply nutrients to plants.
The organic part of the solid phase is formed from the decomposition of organic materials ( plant and animal remains). This part release nutrients into the solution as decomposition takes place . The organic matter has good water holding capacity, bind soil particles together, source of humus, serve as source of food to microorganisms and it is very good in improving poor soil like sand and clay soils etc.
THE SOIL SOLUTION ( LIQUID PHASE)
The soil solution is all liquid water within the soil and the solutes dissolved within it. Solutes include ions, as well as neutrally charged species like sugars and select acids and bases. Gases such as carbon dioxide (CO2) and oxygen (O2) can also dissolve into the solution and react with other solutes, as well as water. During the dissolution process, each molecule of solute is surrounded by water molecules, affecting the behavior of the overall solution. For example, when table salt (NaCl) dissolves, the compound dissociate into Na+ and Cl-. Each of the Na+ ion formed is surrounded by a sphere of water, and likewise the Cl–.
In soil, which is the focus point, solid mineral salts like CaCO3, CaSO4 and NaCl are relatively soluble and therefore dissolve readily. They become part of the soil solution in most soils. The concentration of these salts vary widely in the soil due to the soil mineral composition and formation factors.
Gases like the greenhouse gases ( carbon dioxide (CO2), methane (CH4), and nitrous oxide (N2O)), released from microbial metabolism, are spread everywhere in the soils.
Water is needed for this release. It is an important substance needed in the soil especially for most reactions and assist in nutrient dissolution. In the water solution, majority of soil reactions takes place. Thus, making soil solution properties an important factor that affect soil reactions.
The three most important soil solution properties with respect to this effects are: pH, redox status, and electrical conductivity.
Soil pH: Measures the soil acidity and basicity. It is calculated as the negative log of the hydrogen ion concentration:
pH = -log[H+]
a. REDOX STATUS: Redox status also called oxidation-reduction status, refers to the degree to which a soil environment favours the gain or loss of electrons, reflecting the balance between oxidizing and reducing agents. This status influences lots of soil processes, including; nutrient availability, microbial activity, and the overall health and functioning of the ecosystem.
EFFECTS OF REDOX STATUS ON SOIL PROCESSES
Some of the effects of redox status on soil processes include;
i. NUTRIENT AVAILABILITY:
Redox potential influences the solubility and mobility of various nutrients. For example, in reducing conditions, iron and manganese can become more soluble and mobile, potentially leading to their depletion from certain soil horizons.
ii. MICROBIAL ACTIVITY:
Redox status directly affects the types and activities of microorganisms in the soil. Different microbial groups have specific redox requirements for their metabolic processes. For example, under aerobic respiration, soil microorganisms use oxygen as a terminal electron acceptor in the decomposition of organic matter, releasing carbon dioxide. Also, in very reducing conditions, certain bacteria can produce methane (CH4) from organic matter in a process called “Methanogenesis”.
iii. SOIL STRUCTURE AND COLOUR:
Redox processes can alter soil structure and colour through the oxidation and reduction of iron and manganese compounds, creating visible redoximorphic features like mottles or gleying.
In the case of soil structure,
redox status significantly influence the solubility and precipitation of iron and manganese oxides, which act as cementing agents. That is, under a well-aerated soils, the presence of oxygen promote the formation and precipitation of iron and manganese oxides. These oxides can act as binding agents, creating stable aggregates and improving soil structure.
Conversely, reducing conditions like waterlogged or saturated soils, decreases oxygen availability in the soil. Iron and manganese oxides are reduced to their soluble forms, weakening the soil structure and potentially leading to its degradation.
Additionally, redox potential affects microbial activity, which also plays a role in soil structure development. It influence the types of microorganisms that can thrive in a soil.
Different microbial communities are adapted to different redox conditions, and their activity can affect soil structure. For example, some microorganisms can break down organic matter, which can contribute to soil aggregation, while others may produce substances that can hinder aggregation.
In the case of soil colour, redox conditions can influence soil colour. Oxidized soils often exhibit reddish or yellowish hues due to the presence of oxidized iron compounds. While reduced soils may be gray or greenish due to the presence of reduced iron.
iv. SOIL HEALTH:
The overall redox status of a soil is a key indicator of soil health, influencing its ability to support plant growth and ecosystem functioning.
Redox potential affect the availability of nutrients in the soil, with some nutrients being more available under oxidizing conditions and others under reducing conditions. This can indirectly influence soil structure, nutrient availability, affect plant growth and determine the amount of organic matter in the soil. Thus, determining the healthiness of the soil.
REDOX AND TERMINOLOGIES IN REDOX
a. Redox status measures the redox potential (Eh), [O2], or [CO2]. It reflects the balance between oxidation and reduction reactions within the soil. Redox reactions are greatly influenced by the amount of oxygen and other oxidizing agents in soil such as nitrate.

b. Soil redox potential (Eh) is a measure of the electrical potential of soil due to the tendency of the chemicals within it to donate or accept electrons. It indicate the relative abundance of oxidized and reduced substances. It reflects the balance between oxidation and reduction reactions, primarily influenced by oxygen availability and microbial activity.
In well-aerated soils, oxygen acts as a primary electron acceptor, resulting in a higher redox potential. This therefore makes the more important nutrients like nitrogen and iron to become oxidized by donating an electron to oxygen.
Conversely, in waterlogged or poorly drained soils, oxygen is limited, and other electron acceptors (like iron, manganese, and nitrogen) are utilized, leading to a lower redox potential. This Low redox potential can negatively impact plant growth by inhibiting respiration and producing toxins under reducing conditions.
IMPORTANCE OF REDOX POTENCIAL
1. Redox potential plays a critical role in both agricultural and horticultural systems, influencing nutrient availability, microbial activity, and overall soil health.
2. Additionally, soil redox dynamics are fundamental in shaping natural ecosystems, often delineating boundaries between different vegetation communities such as wetlands, grasslands, and forests.
HOW TO MEASURE REDOX
Scientists measure soil redox potential using the Eh scale (measured in millivolts):
i. HIGH Eh VALUES (+400 to +700 mV): Well-oxygenated, oxidized conditions
ii. LOW Eh VALUES (-300 to +200 mV): Oxygen-depleted, reduced conditions
CLASSES OF SOIL REDOX POTENCIAL
Kaurichev and Shishova (1967) defined four classes of soil redox conditions as follows:
i: Eh > +400 mV -> aerated soils.
ii: Eh +100 to +400 mV -> moderately unaerated (reduced) soils.
iii. : Eh -100 to +100 mV -> strongly unaerated (reduced) soils.
iv: Eh -300 to -100 mV -> anaerobic, or highly reduced, soils.
For agricultural soils, Eh values between 400 and 500 mV are highly favourable conditions (Husson 2013).
INSTRUMENTS USED IN MEASURING SOIL REDOX
i. Redox potential (Eh), can be measured using a portable, handheld meters which are quicker and easy spot measuring instrument.
ii. SWAP Instruments redox probes: This is an instrument that has an integrated reference probe for easy and efficient installations. The equipment is also suitable for installation in many settings, including pots in a glasshouse, soil columns in a laboratory, or field conditions such as wetlands, peat, forests, woodland, agriculture, and horticulture
iii. Redox potential can also be measured using an electrode (often platinum) inserted into the soil, often in a soil-water suspension with a reference electrode (like Ag/AgCl) used to establish a potential difference.
RANGE OF REDOX POTENCIAL IN SOILS
Redox potential in soils can vary significantly, with upland soils typically having higher redox potentials (above +300 mV) and wetland soils or flooded areas exhibiting lower potentials (potentially below +300 mV or even negative values).
c. ELECTRICAL CONDUCTIVITY (EC): EC is a measure of the soil’s ability to conduct electricity, which is largely determined by soil salinity. That is, concentration of dissolved salts in the soil water. Higher EC indicates a greater concentration of salts, which can influence various soil properties and processes. Or EC can be said to be a measure of how easily electrons can flow through soil.
EC is directly proportional to the total concentration of ions in solution, and thus EC is a good measure of salinity or soil “saltiness.”
Salinity affects numerous soil properties such as aggregation, infiltration, and drainage. It also affects soil osmotic potential and water uptake by plants.
The total ion concentration in soil also affects reaction rates in the soil.
Soil redox status significantly influences electrical conductivity. Under reduced conditions like waterlogged, flooded or anaerobic soils, microorganisms break down organic matter, releasing ions like iron (Fe) and manganese (Mn) into the soil solution. These ions coupled with lower redox potential (Eh), along with other dissolved salts, contribute to increased electrical conductivity.
Conversely, oxidized conditions, that is, aerobic conditions, the dissolved ions are oxidized and precipitated out of the solution. Thus, decreasing the number of charge carriers and thus decrease electrical conductivity.

SUMMARY OF THE RELATIONSHIP BETWEEN REDOX POTENTIAL (Eh) AND ELECTRICAL CONDUCTIVITY (EC):
a. LOWER Eh (Reduced):
A lower redox potential (more reducing conditions) generally corresponds to higher electrical conductivity due to the increased presence of dissolved ions.
b. HIGHER Eh (Oxidized):
A higher redox potential (more oxidizing conditions) generally corresponds to lower electrical conductivity as ions precipitate out of solution.
FACTORS INFLUENCING THE RELATIONSHIP
Certain factors influence the relationship between Redox Potential and electrical conductivity. Such factors include; Soil moisture level, soil organic matter decomposition rate, soil texture and structure,
microbial activity and
temperature.
i. SOIL MOISTURE LEVEL: Soil moisture acts as a critical link between redox potential and electrical conductivity. It significantly influences the relationship between redox potential (Eh) and electrical conductivity (EC) in soil. Higher moisture levels generally lead to lower redox potentials and higher electrical conductivity because water acts as a medium for electron transfer, facilitating redox reactions, and also provides a pathway for electrical current to flow. When soil moisture is increased, this leads to higher electrical conductivity because water acts as a solvent for ions, and the more water there is, the more ions can move and carry an electrical charge. With regards to this, when soil moisture is increased and the soil becomes saturated, oxygen diffusion is limited, creating anaerobic conditions. This favours reduction reactions, leading to lower (more negative) redox potentials.
ii. SOIL ORGANIC MATTER DECOMPOSITION:
Organic matter is a source of electrons when it decomposes. Soil organic matter (SOM) decomposition significantly influences the relationships between redox potential (Eh) and electrical conductivity (EC) in soil. Increased SOM decomposition leads to a decrease in redox potential (more reducing conditions) and can increase EC by releasing ions into the soil solution.
A lower Eh indicates a more reducing environment, where oxygen is scarce and other electron acceptors (like nitrate, iron, and manganese) are used by microbes. The decomposition of SOM releases various ions (cations and anions) which are electron acceptors, into the soil solution. These electron acceptors (like iron and manganese oxides) are then reduced, leading to a decrease in redox potential due to the consumption of oxygen and the reduction of other electron acceptors.
In aerobic (oxygen-rich) conditions, oxygen acts as the primary electron acceptor, and organic matter is oxidized to CO2 and H2O.
The ions also contribute to the overall ionic strength and increase the soil’s electrical conductivity. As the soil organic matter decomposition rate increases, this leads to increased concentrations of dissolved ions, resulting in a higher EC.
For example, the addition of organic matter like manure or compost can increase EC due to the release of cations and anions.
iii. SOIL TEXTURE AND STRUCTURE:
Soil structure and texture significantly influence both redox potential and electrical conductivity, affecting how these properties interact within the soil. Soil structure, referring to the arrangement of soil particles, impacts aeration and water movement, which in turn affects redox potential. Soil texture, determined by the proportions of sand, silt, and clay, influences the surface area available for reactions and the movement of ions, affecting both redox and electrical conductivity.
A good structure (e.g., granular or crumb structure or sandy soils) possesses larger pores, thus, promoting better aeration and drainage. This can lead to more oxidized conditions (higher redox potential) as oxygen can readily reach microbial communities, influencing organic matter decomposition and nutrient cycling. Also, such soils typically have lower EC due to the larger pore spaces and lower CEC, resulting in fewer dissolved ions.
On the other hand, poor structured soils (e.g., compacted or massive structure) such as clay soils, especially under waterlogged conditions, restricts aeration and drainage, creating anaerobic (oxygen-limited) conditions. This can lead to reduced redox potentials as microbes utilize alternative electron acceptors like nitrates, sulfates, and iron oxides. Also, such soils generally have higher EC. Clay minerals, especially those with high cation exchange capacity (CEC), contribute to the presence of ions in the soil solution, increasing conductivity.
iv. MICROBIAL ACTIVITIES;
Microbial activity significantly influences the relationship between redox potential and electrical conductivity.
Some of the functions of microbes is that microbial activity influences the concentration of ions by accelerating the weathering of minerals, thus, releasing ions into the surrounding environment, thereby increasing conductivity. They also assist in the decomposition of organic matter, thus, release ions and affect conductivity. And lastly, microbial processes can alter pH, which in turn can affect the solubility and mobility of various ions, thus influencing conductivity.
Redox potential, which indicates the tendency of a substance to gain or lose electrons, is directly affected by microbial respiration and metabolism, as microbes utilize electron donors and acceptors in their processes. These microbial reactions, in turn, impact the availability of charged ions and molecules, which are key determinants of electrical conductivity.
v. SOIL TEMPERATURE:
Soil temperature significantly influences both redox potential and electrical conductivity, with higher temperatures generally increasing both parameters. Temperature affects the rate of redox reactions, impacting the availability of electrons and ions involved in these reactions, thus influencing redox potential. Simultaneously, higher temperatures increase the mobility of ions in the soil solution, leading to a rise in electrical conductivity.
Soil temperature generally influence the chemical and biological processes taking place in the soil.
Higher temperatures for example, accelerate chemical and biological reactions in the soil, including those involved in redox processes.
Redox reactions involve the transfer of electrons, and their rates are temperature-dependent. For example, a common redox reaction involving iron (Fe³⁺/Fe²⁺) is affected by temperature, with a 1°C increase resulting in a small change in the measured potential.
Soil temperature also influences microbial activities which play a crucial role in redox processes. Increased temperature can stimulate microbial activity, affecting the rates of oxidation and reduction reactions.
When ions are releases into the soil, in the presence of
temperature, the mobility of these ions in the soil solution are directly affected. As the soil temperature increases, the kinetic energy of the ions also increase, making them move more freely and increasing electrical conductivity.
d. SOIL SURFACES: The soil surface is also refered to as the Solid-Solution Interface.
Solids have very slow reaction rates, therefore, the vast majority of reactions with solids occur at the solid-solution interface, that is, the surfaces of soil particles where they meet soil water. These reactions include dissolution of the solids into the solution, precipitation of new solids onto the solution, and adsorption of solutes onto surfaces. (This is one of the reasons why particle size and surface area are so important in soil science).
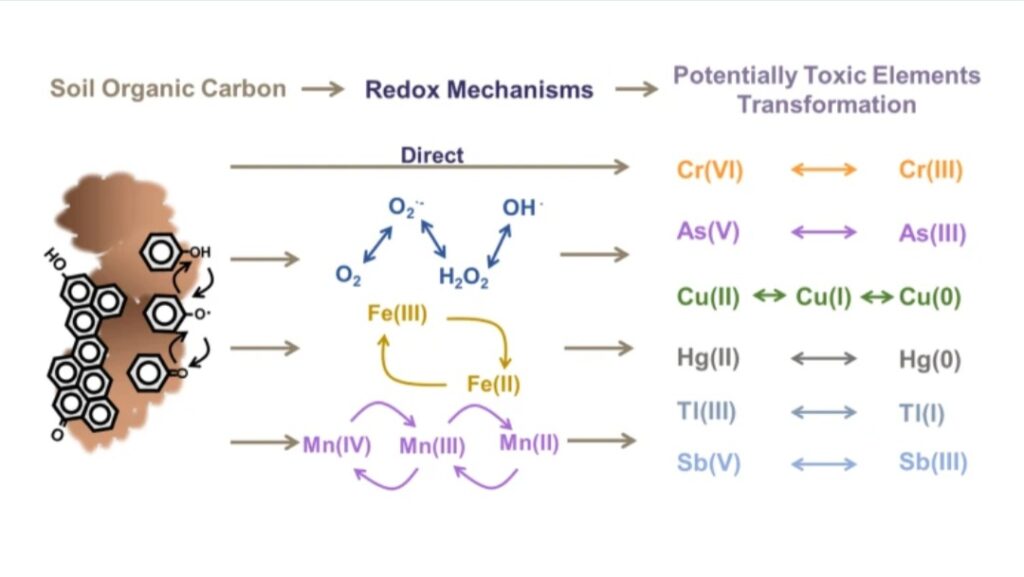
Redox status significantly impacts the soil solid-solution interface by influencing the solubility, mobility, and bioavailability of various elements, including nutrients and contaminants. Changes in redox potential alter the chemical forms (speciation) of these elements, affecting their interactions with soil particles and their availability for plant uptake or microbial activity.
Adsorption which can simply be described as “stickiness” or adherence of solutes to solid particle surfaces, can also be used to describe how nutrients and contaminants are adsorbed to soil particle surfaces, enabling soil to retain nutrients for plants while keeping contaminants out of the groundwater. Thus, it is said that soil surfaces support three important soil ecosystem services:
i. supporting plant growth,
ii. providing nutrients, and
iii. filtering water.
RELATIONSHIP BETWEEN REDOX POTENTIAL AND THE ENVIRONMENT
a. SOIL REDOX AND MICROBES
Soil redox potential has a strong relationship with soil microbial communities. Each group of microorganisms thrives within specific Eh and pH ranges. High Eh environment is favourable for the survival of aerobic bacteria and actinomycetes. On the other hand, low Eh environment is favourable for the survival of anaerobic bacteria such as denitrifiers, sulfate-reducers, and methanogens. These microbes plat important roles in nutrient cycles, including nitrification, denitrification, and methane production. Likewise, they also help in improving soil fertility and ecological balance.
b. SOIL REDOX AND WATERLOGGED SOILS
Redox potential is an important factor in wetland and saturated soils. Here, water saturation dominate the environment during the wet season, thus, creating an anaerobic conditions, while in the dry season, the water level becomes lowered, and thus, an aerobic condition takes place within the soil. This brings about a fluctuation between the two seasons. Such fluctuations can have dramatic effects on nutrient cycling, greenhouse gas emissions, and plant root health. For instance, prolonged reducing conditions can lead to the accumulation of toxic compounds such as hydrogen sulfide (H₂S) or reduced iron (Fe²⁺), which may inhibit plant growth.
c. SOIL REDOX AND SOIL pH: Soil pH and redox potential (Eh) are interconnected and influence each other through various chemical and biological processes in the soil. Most scientists give more preference to soil pH than redox. While pH is a measure of acidity or alkalinity, redox potential indicates the soil’s capacity to gain or lose electrons, and these two are linked because many redox reactions involve the transfer of hydrogen ions (which determine pH). For example, the oxidation of ferrous iron (Fe2+) to ferric iron (Fe3+) consumes hydrogen ions, potentially increasing soil pH, while the reduction of nitrate (NO3-) to ammonia (NH4+) releases hydrogen ions, or sulfate to sulfide typically alters the pH of the surrounding soil microenvironment. Conversely, it should be noted that this relationship is vice versa. The pH of the soil inversely influence the solubility of various chemical compounds, including those involved in redox reactions. For instance, the solubility of iron and manganese oxides, important components in redox processes, is pH-dependent, with lower pH values generally favouring their solubility. Therefore, understanding and managing soil Eh and pH together provides a more comprehensive concept of soil chemistry and biological activity.
d. SOIL REDOX AND PLANT GROWTH : Soil redox potential (Eh) significantly impacts plant growth by influencing nutrient availability, oxygen levels, and the presence of toxic compounds. Optimal redox conditions promote healthy plant development, while extreme values can be detrimental. For example, a well-aerated (oxidized) soils generally support healthy plant growth due to increased oxygen availability and nutrient solubility. Essential nutrients like nitrogen and phosphorus becomes more soluble and generally become more available to plants, while poorly aerated (reduced or low redox) soils can hinder growth due to oxygen deficiency and the production of toxic substances. Some nutrients become converted into forms that are less accessible or even toxic to plants. Apart from affecting nutrient availability, plant roots for example, influence soil Eh through root respiration, organic exudates, and oxygen transport. Wetland plants like rice are oxygenate their rhizosphere, raising the redox potential and mitigating the toxicity of reduced compounds. Conversely, in aerobic soils, plant roots and associated microbes can lower Eh by consuming oxygen and releasing reducing compounds. This rhizosphere-mediated modulation of redox conditions is essential for maintaining nutrient availability and promoting healthy root-microbe interactions.
e. SOIL REDOX AND SOIL FORMATION : Soil redox potential (Eh) is an important factor in soil formation. It influences various soil formation processes like organic matter decomposition, nutrient availability, and mineral transformations. Redox reactions, which involve the transfer of electrons, are heavily impacted by the availability of oxygen in the soil. This, in turn, affects the types of microorganisms present and the overall health and characteristics of the soil. Eh influences soil formation processes (pedogenesis). It affects how quickly organic matter decomposes especially in aerobic conditions where decomposition is faster, thus, releasing nutrients. Under anaerobic conditions, decomposition is slower, and some organic matter may be preserved. It can mobilize or immobilize nutrients. For example, iron and manganese can exist in different oxidation states, affecting their availability to plants. It can also cause the mobility of environmental pollutants such as arsenic, mercury, and cadmium. It also governs the production and consumption of greenhouse gases like methane (CH₄) and nitrous oxide (N₂O), linking soil redox status to broader climate and environmental concern.Lastly, redox reactions can alter the composition of minerals in the soil. It can impact its structure and properties. For instance, the reduction of iron oxides can lead to the formation of different mineral phases.
